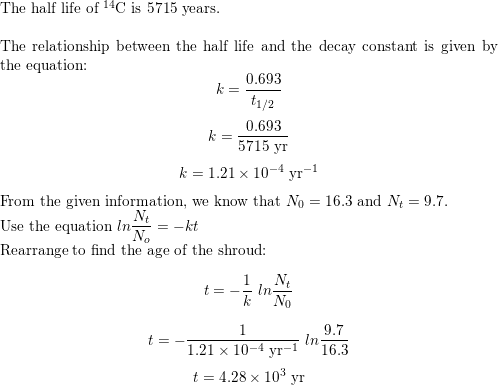SOLVED: The number of disintegrations per minute (dsm) of a sample of iodine-133 is measured at 5.5 × 102 dsm. When the sample is measured 295 minutes later, the number of disintegrations

Calculations and Instrumentation used for Radioligand Binding Assays - Assay Guidance Manual - NCBI Bookshelf

The disintegration rate of a certain radioactive sample at any instant is 4750 disintegrations - YouTube
How to find disintegration mass after 2 days if the half life period of radioactive substances is 4 days - Quora

Calculate the number of disintegrations per second of 1 g of a radioactive sample whose half-life is - YouTube

SOLVED: A sample of a radioactive isotope decays at a rate of 3.2 10* disintegrations per minute initially and 4.1 103 disintegrations per minute after 3 weeks What is the half-life of
17.6: Radiocarbon Dating: Using Radioactivity to Measure the Age of Fossils and Other Artifacts - Chemistry LibreTexts

The rate of disintegration was observed to be 1017 disintegrations per sec when its half life period is 1445 years. The original number of particles are.

The disintegration rate of a certain radioactive sample at any instant is 4750 disintegrations per minute. Five minutes later the rate becomes 2700 per minute. Calculate (a) decay constant and (b) half-life

A radioactive element has rate of disintegration 10,000 disintegrations per minute at a particular instant. After four minutes it becomes 2500 disintegrations per minute. The decay constant per minute is

SOLVED:(II) A sample of ^23392U (T(1)/(2) = 1.59 ×10^5 yr) contains 4.50 ×10^18 nuclei. (a) What is the decay constant? (b) Approximately how many disintegrations will occur per minute?